 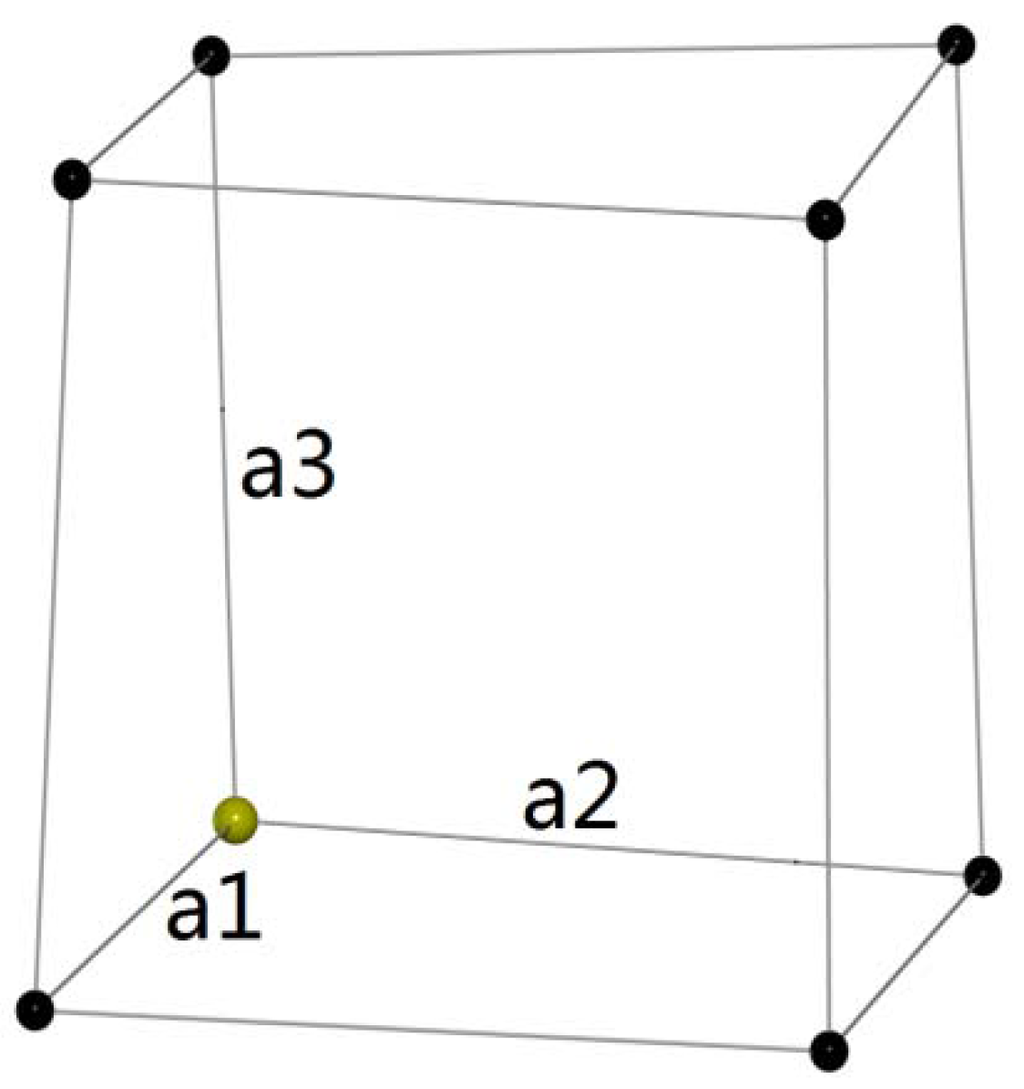
The choice of the particular way to evaluate if a given material should be considered a “high entropy” oxide is troublesome. 6 This is a particular point of contention as we explore the paradigms of defining the “level of entropy” in a given composition both from a theoretical and an experimental point of view. Moreover, the short-range disorder competes with short-range order as it is observed both in high entropy metal–metal bonded alloys 5 and ionically and covalently bonded oxides. These depend on the valence states, electronegativities, preferred coordination numbers, ionic radii, and masses of particular cations and is a feature that may be controlled only in a very limited way. The short-range disorder arising from the compositional complexity influences functional properties of materials, such as their thermal and thermoelectric, 1 dielectric, 2 magnetic, 3,4 and many other properties. 
The uniqueness of this wide group of materials is that they show a long-range structural crystalline order and short-range compositional disorder. The number of variables influencing functional properties in this field is enormous. In essence, the entropy term might be considered as a controllable order parameter that can provide a route to designing highly tunable properties and novel emergent functionalities. The key to utilizing high entropy materials for desirable applications is the ability to exploit novel functional structure–property relationships by manipulating entropy to form unique crystal structures, valence states, coordination numbers, and local distortions in the lattice. Conceptually, a system’s entropy might be viewed as a bank, in which entropy can either be deposited to randomize structure or withdrawn to stabilize materials with chemical, structural, or charge ordering within the high entropy matrix. Ideal entropy can be considered as the value of the entropy in the limit as the system becomes fully disordered-as ordering increases, the system’s entropy value reduces away from this ideal value. Utilizing configurational entropy as the thermodynamic driver for stable solid solutions allows access to a vast and unexplored materials space that is unattainable with enthalpy stabilized methodologies. Furthermore, the thermodynamic stability during synthesis may limit the ability to realize these functionalities in real materials. Designing and realizing a material having desired functional properties is a difficult task since the chemical composition, morphology, microstructure, stability, functional properties, and synthesis are not independent. 
For simplicity, controlling materials relies on what elements are in the material and how those elements’ nearest neighbor environments influence electron interactions. The second way of influencing material properties is by choosing the most appropriate morphology and micro- or nanostructure, which may be realized using various synthesis techniques. The first one is a chemical modification through substitution of the host cations with others chosen based on their electronic configuration, ionic radius, and other basic features. Simplifying this, there are two main conventional ways of tailoring the functional properties of oxides. The functional properties of metal oxides constitute one of the widest research fields belonging to many fundamental and applied scientific disciplines, e.g., materials science, chemistry, physics, engineering, and many others. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
